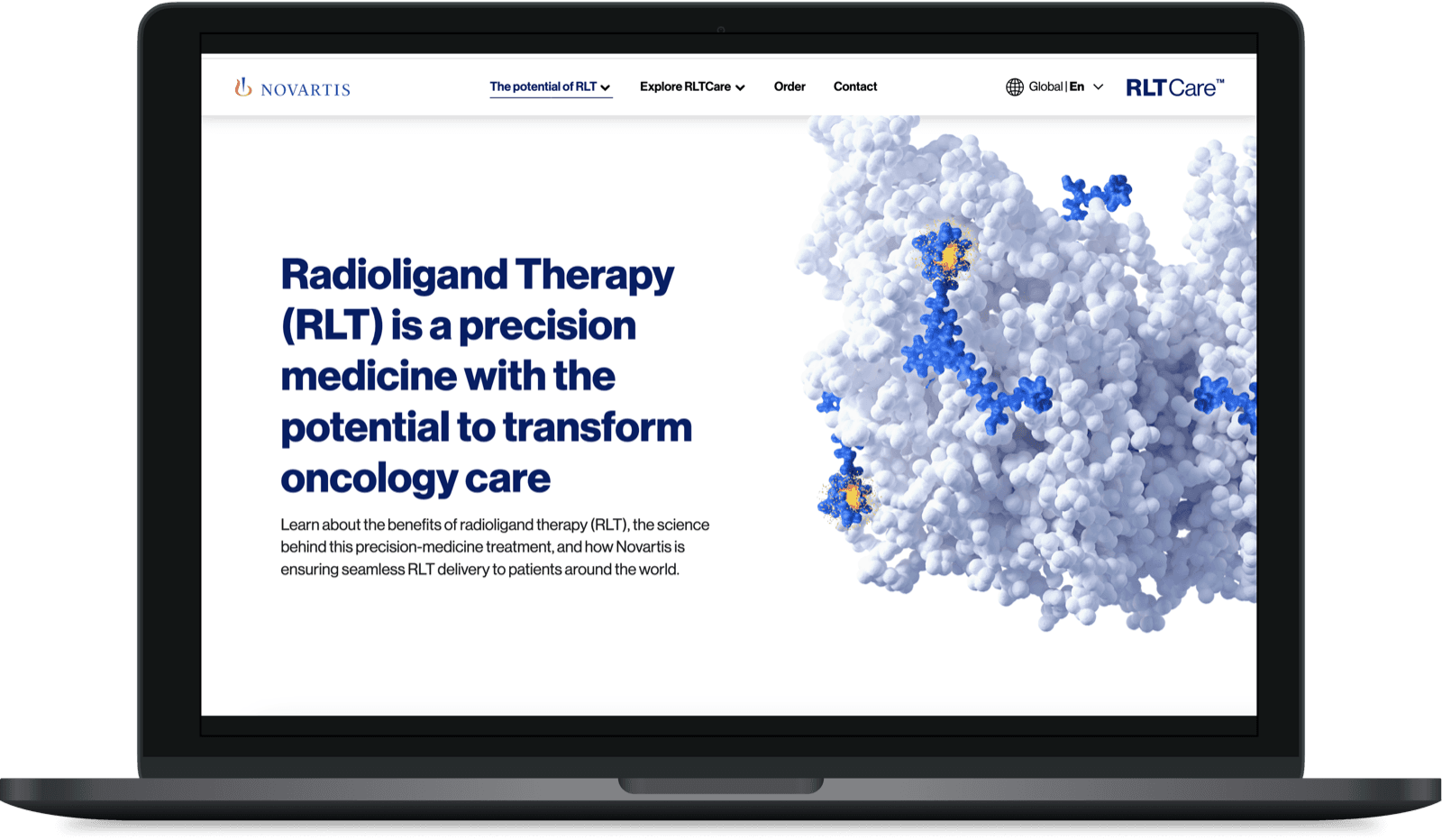
Novartis RLT Care
Leading content strategy and design for Novartis' first global Radioligand Therapy hub, turning scattered resources into a unified platform for healthcare professionals.
Client
Novartis
Role
Content design lead
Timeline
2023 to 2025
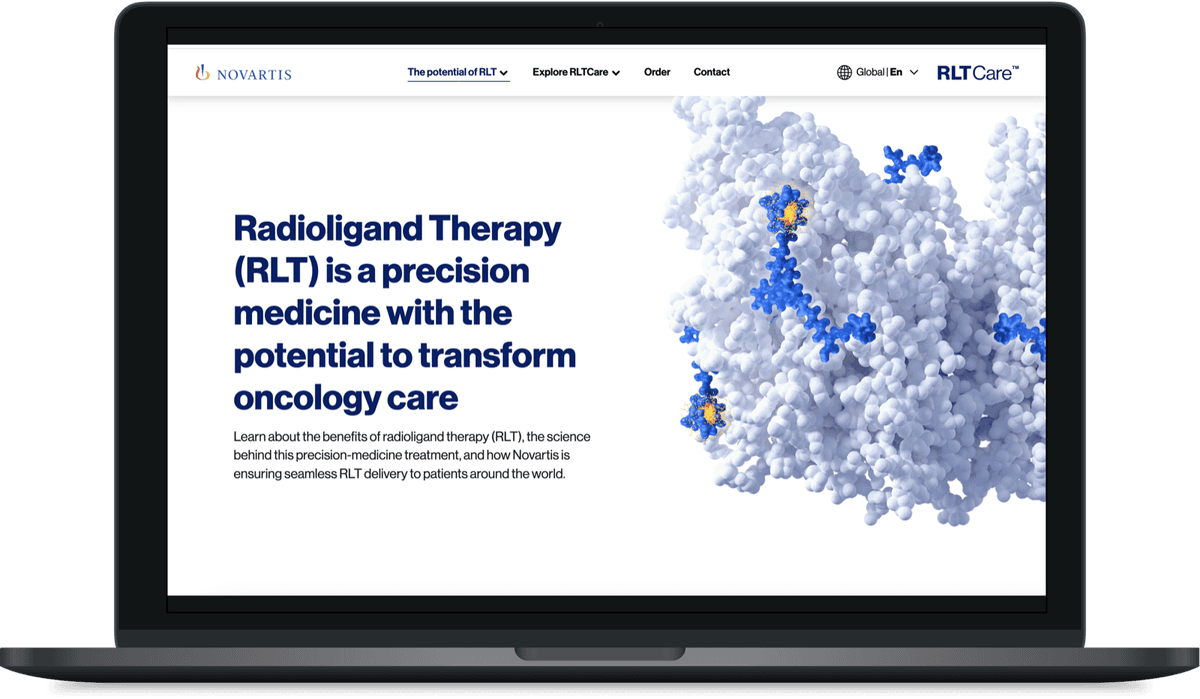
A breakthrough therapy with no digital home
Radioligand Therapy is changing cancer treatment. But when I joined this project, Novartis had no unified digital presence for it.
Content was scattered across PDFs, microsites and disconnected platforms. Healthcare professionals struggled to find what they needed. Local markets were building their own solutions from scratch.
The brief: create a single platform that works globally but adapts locally, while navigating the regulatory complexity of pharma.
Content design lead
I owned the content strategy and design across the entire platform. This meant working at the intersection of medical accuracy, regulatory compliance, and user experience.
Defined the information architecture and content model
Created role-based journeys for different healthcare professionals
Built a modular content system that scales across markets
Collaborated with medical, legal and regional teams
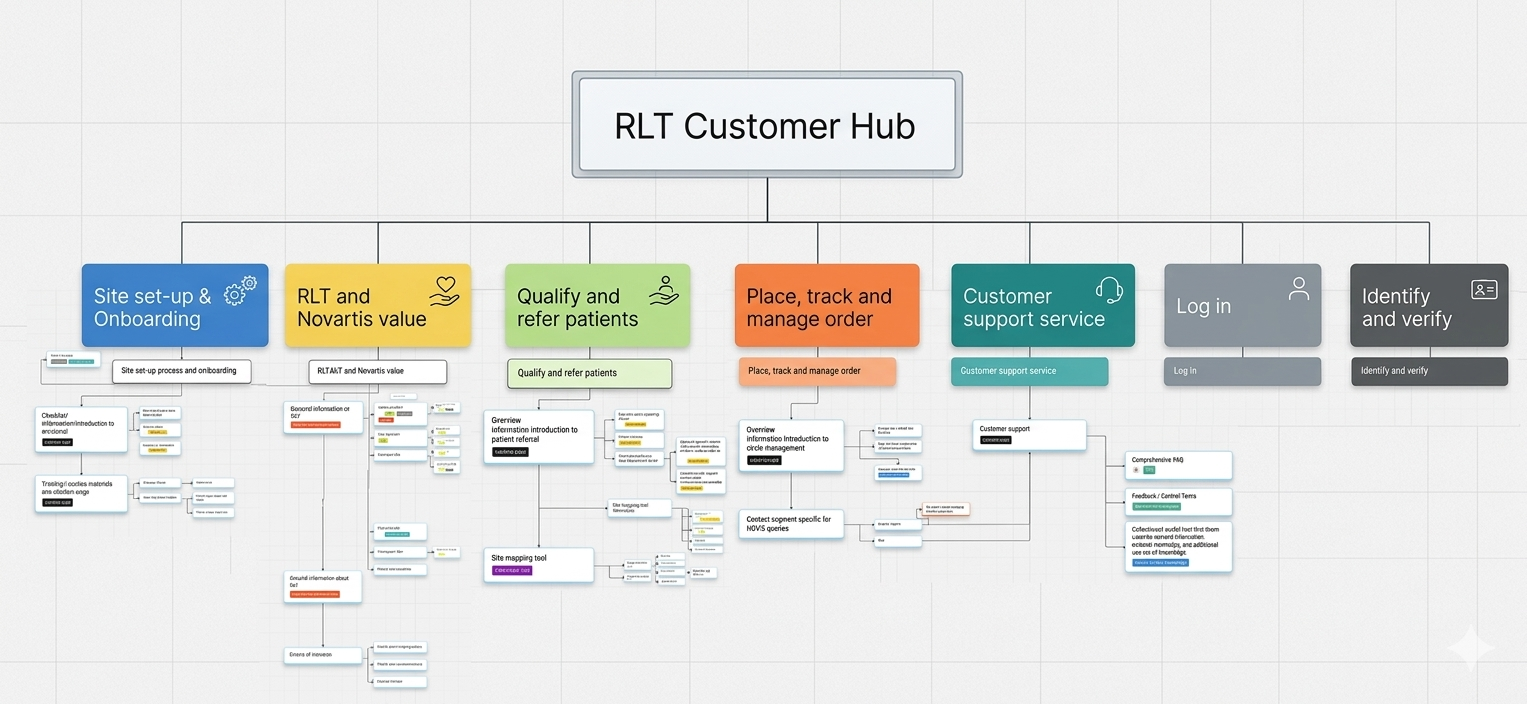
Four distinct user types, one platform
RLT involves a complex care pathway. Different healthcare professionals need different information at different moments. I mapped these needs to design journeys that actually work.
Referring HCPs
Gastroenterologists, haematologists, endocrinologists, uro-oncologists, radiation oncologists, urologists
HCPs managing treatment
Nuclear medicine physicians, radiopharmacists, imaging centres, nurses, care teams, admin teams
Patients and caregivers
Non-medical stakeholders seeking clear, accessible guidance
Novartis teams
Internal sales and commercial stakeholders
Solving real problems
Every project has moments where a decision shapes everything that follows. Here are three that defined this one.
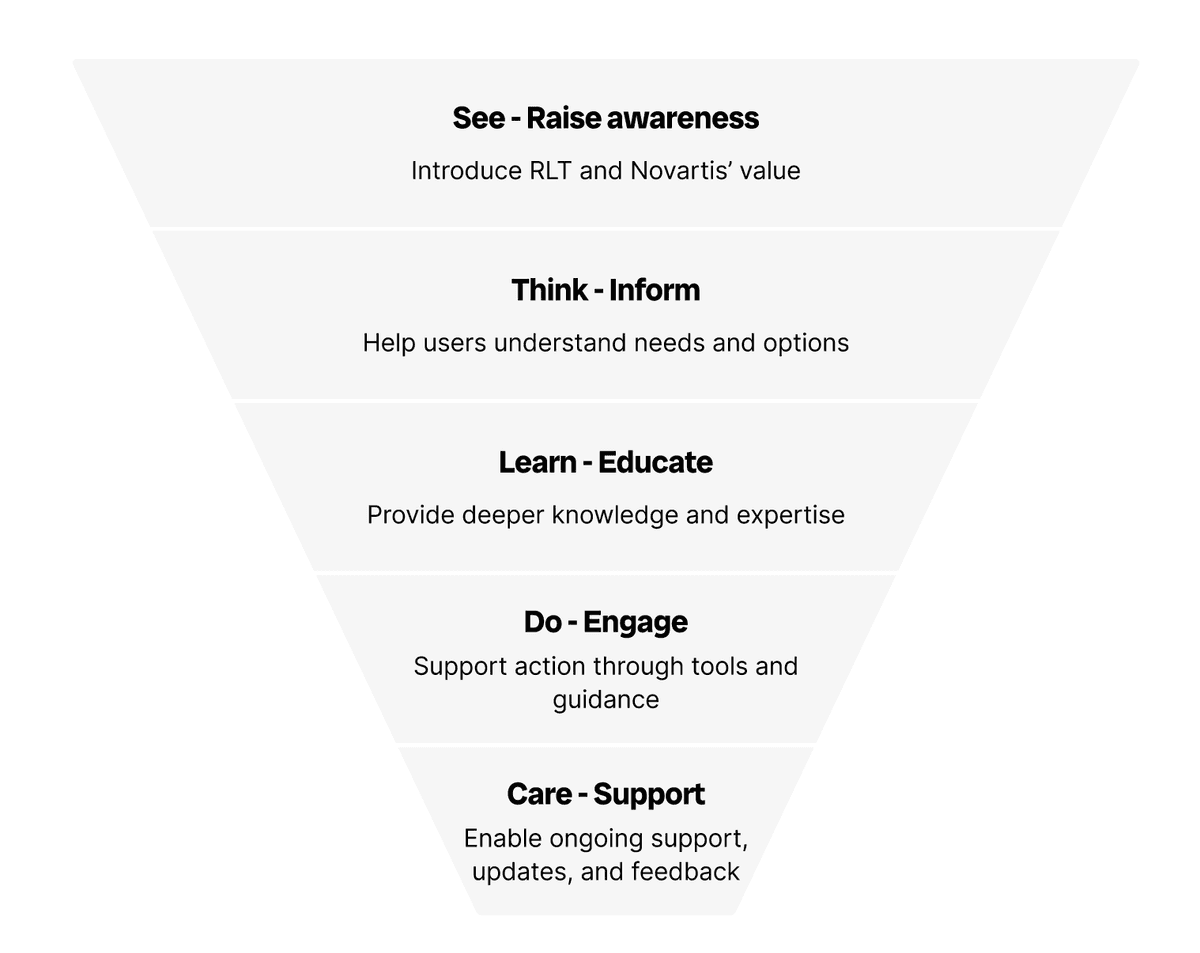
A journey-based content strategy
Rather than organising by product, I mapped content to the user journey. This created a clear strategy that guides HCPs from first awareness through to ongoing support.
- →See - Raise awareness: Introduce RLT and Novartis' value
- →Think - Inform: Help users understand needs and options
- →Learn - Educate: Provide deeper knowledge and expertise
- →Do - Engage: Support action through tools and guidance
- →Care - Support: Enable ongoing support, updates, and feedback
Experience principles as guardrails
With multiple markets, languages and regulatory requirements, consistency was a real risk. I established five experience principles that every piece of content had to meet:
- →Clinically grounded - Always lead with evidence
- →Role-aware - Speak to specific needs
- →Action-oriented - Help people do, not just learn
- →Globally consistent - Same structure, localised content
- →Regulation-ready - Built for MLR approval
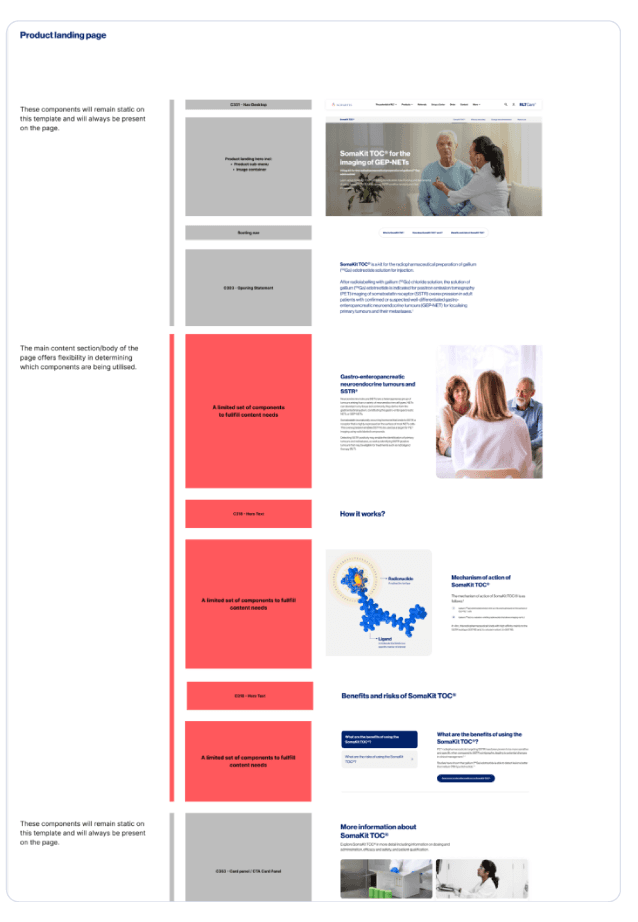
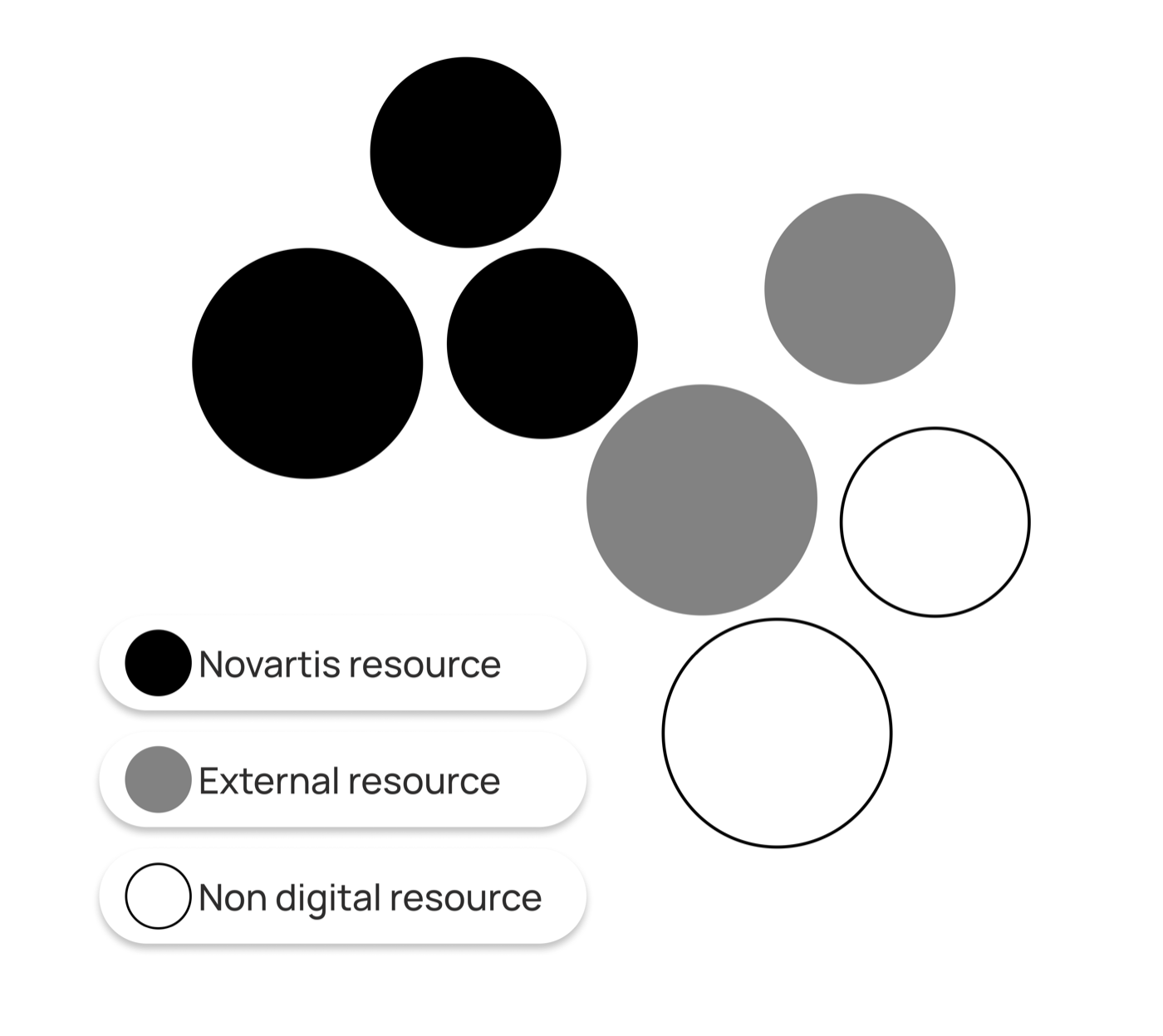
Global framework, local flexibility
Each market has different regulations, approved claims and cultural contexts. A rigid system would break. A completely flexible one would fragment.
I designed a modular content system with three layers:
- →Global core - Locked structure, IA, and key messaging
- →Regional flex - Adaptable claims and evidence
- →Local slots - Market-specific content and resources
This gave markets autonomy within clear boundaries, making rollout faster and governance simpler.
 Global | EN
Global | EN France | FR
France | FR Spain | ES
Spain | ESFrom scattered to scalable
The platform launched and continues to expand. More importantly, the content system I built is now the foundation for how Novartis approaches similar challenges.
8
Markets live
Global reach with local flexibility
1
Unified platform
Replacing scattered PDFs and microsites
Reusable framework
A system Novartis can evolve and reuse
What the team said
Thanks for reading
Explore more of my work or get in touch to discuss your project.